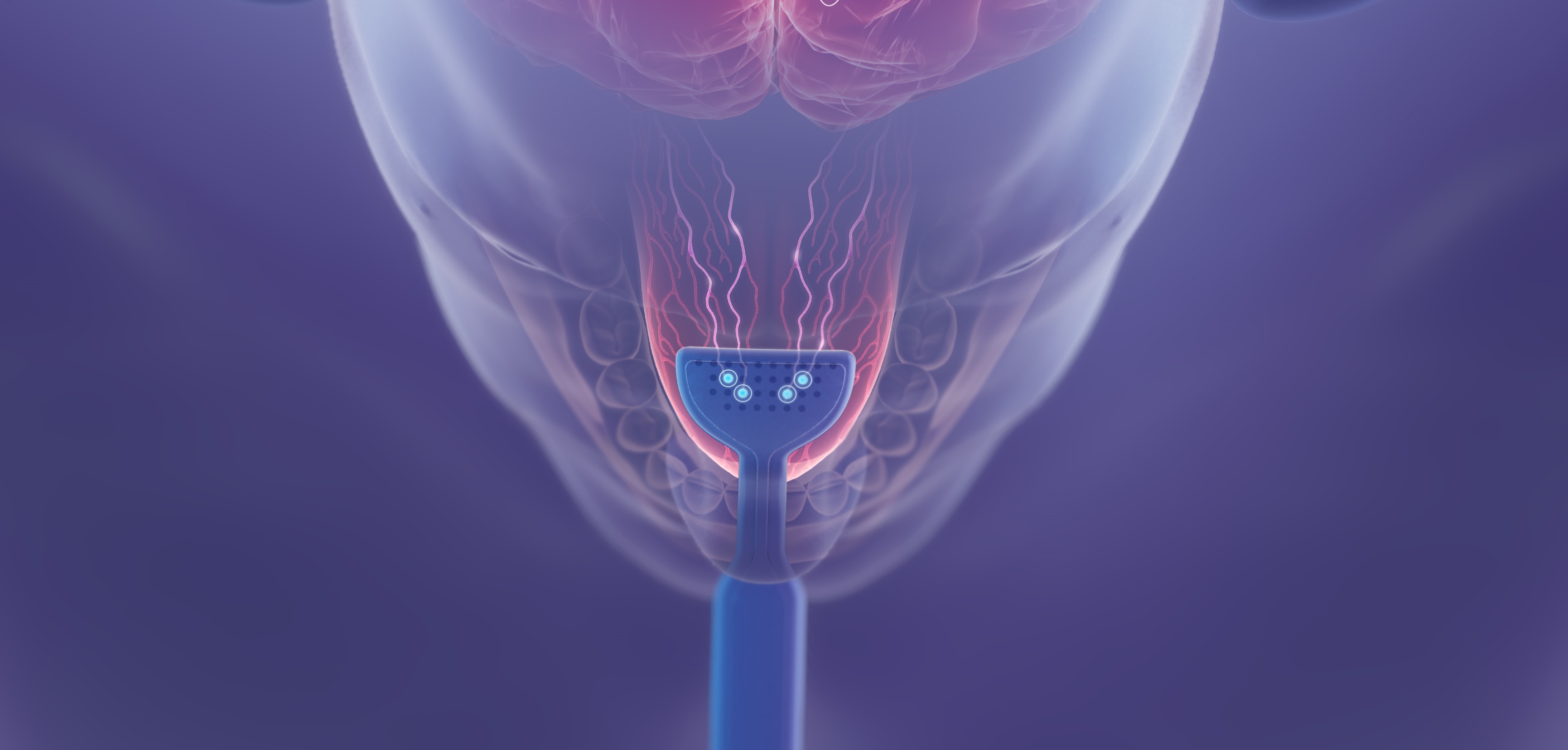
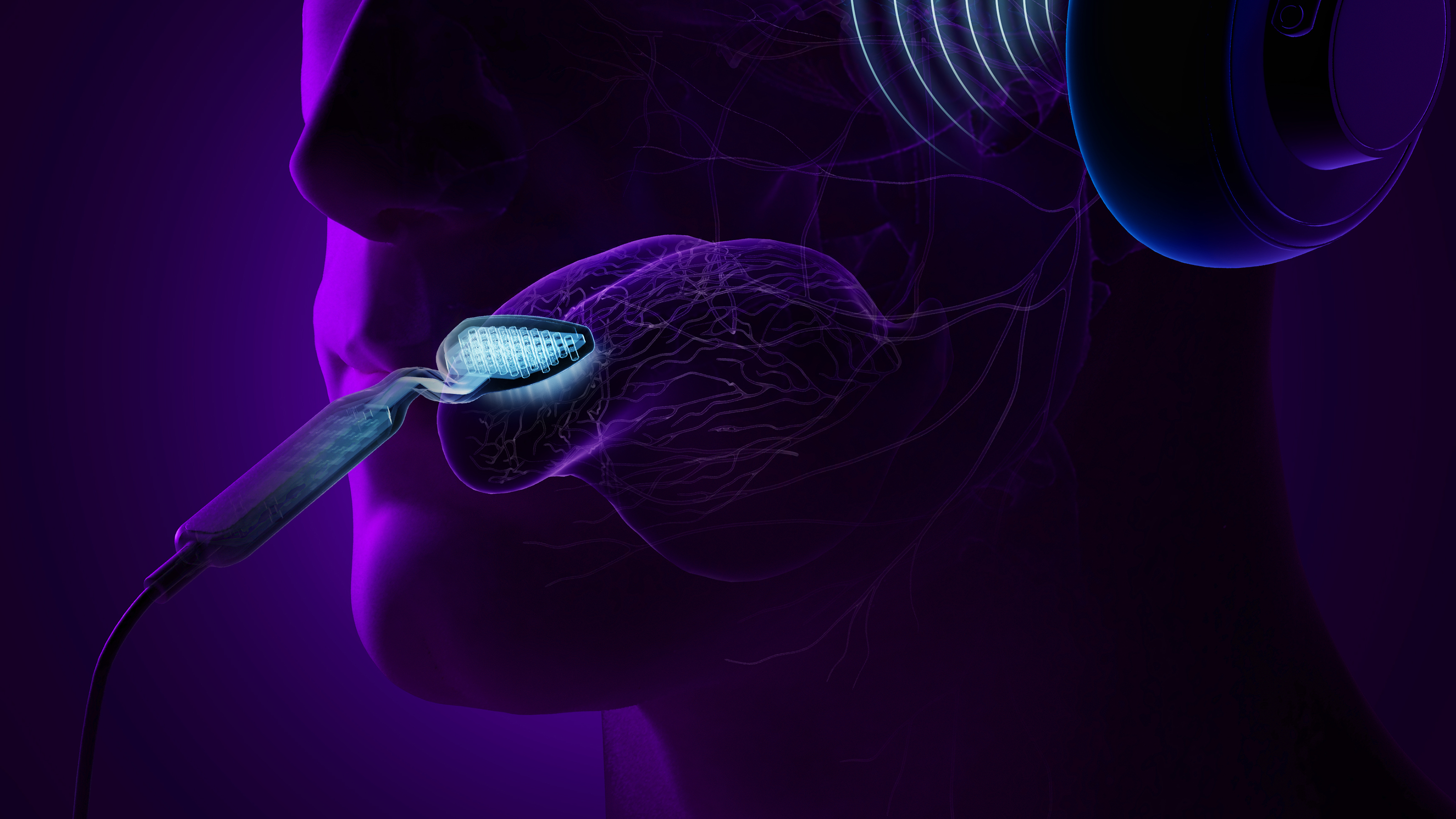
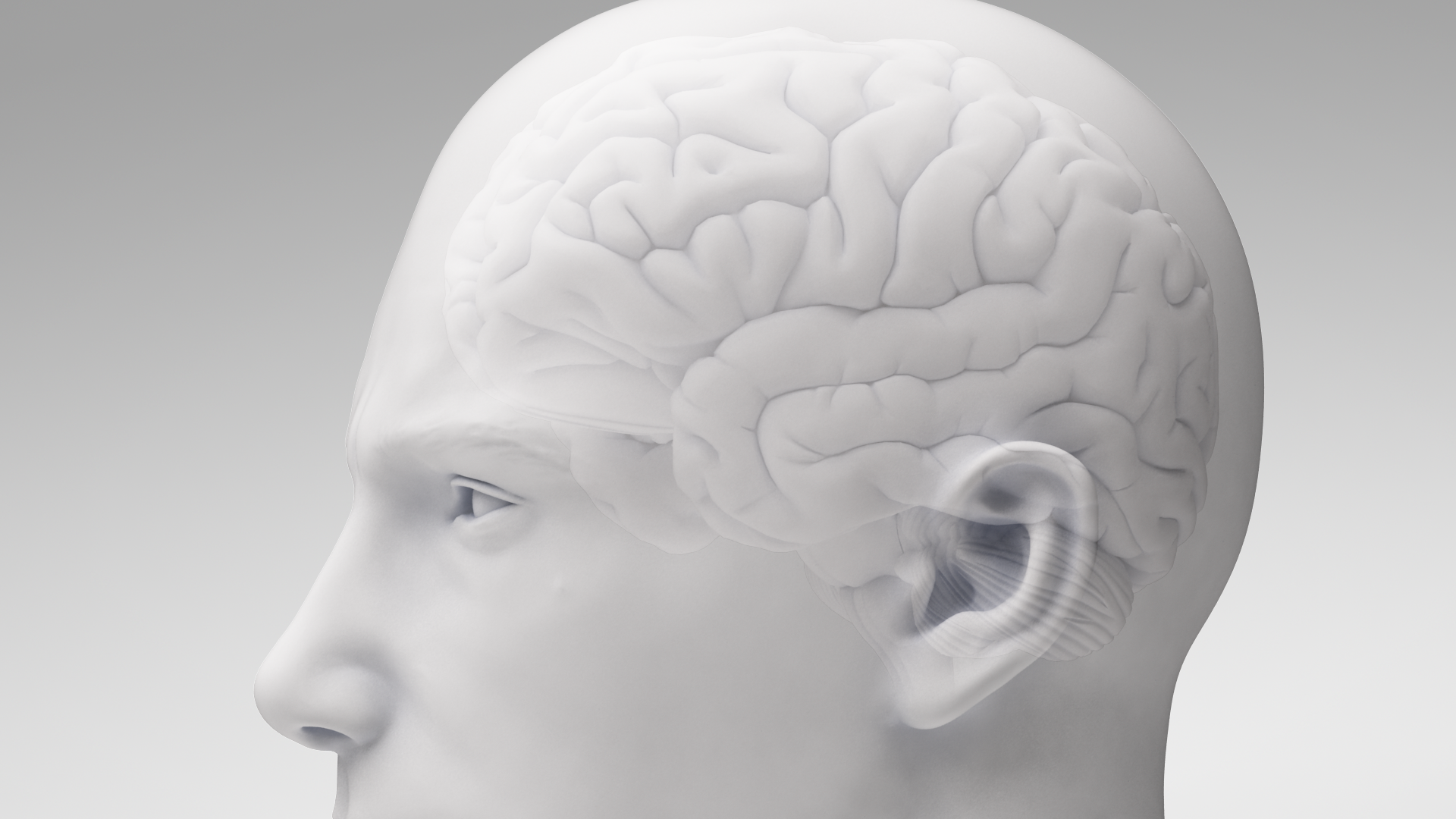
Newsroom
Please find our latest news and upcoming events below. If you are a member of the media and have a media query please contact:
Following an announcement made by the Irish Government on Friday 27th March, restrictions have been put in place and appointments have been rescheduled until further notice.
We continue to provide support to our patients during this time. Services will be delivered in a different way, as face to face appointments will be not offered until further notice. You can contact us via email:
For general queries and tongue tip orders: reception@neuromodmedical.com
For appointments: appointments@neuromodmedical.com
You may also contact us via phone on +353 1 253 1448. Our phone lines remain open Monday to Friday 9am – 5pm (with the exception of bank holidays).
If necessary, a consultation can also be arranged for a particular day & time that suits, either via telephone or video call for;
In some instances, a video call may be a better option, so you can talk to one of our clinical team face to face. You can use your mobile phone, Ipad, tablet or computer. You will need internet or wi-fi access. Please call or email us and we would be happy to advise you on the best option.
Email HereStimulating information
Please find our latest news and upcoming events below. If you are a member of the media and have a media query please contact: